NSE letter: A CAPA plan for your 510k process
Cry, complain, call the reviewer…you might feel a little better, but you received an NSE letter, and tomorrow you still can’t sell your device.

Instead, try approaching an NSE letter like a CAPA investigation. What is the issue? The FDA determined that your device is not substantially equivalent to the predicate you selected. What is the root cause? There are four (4) possible root causes.
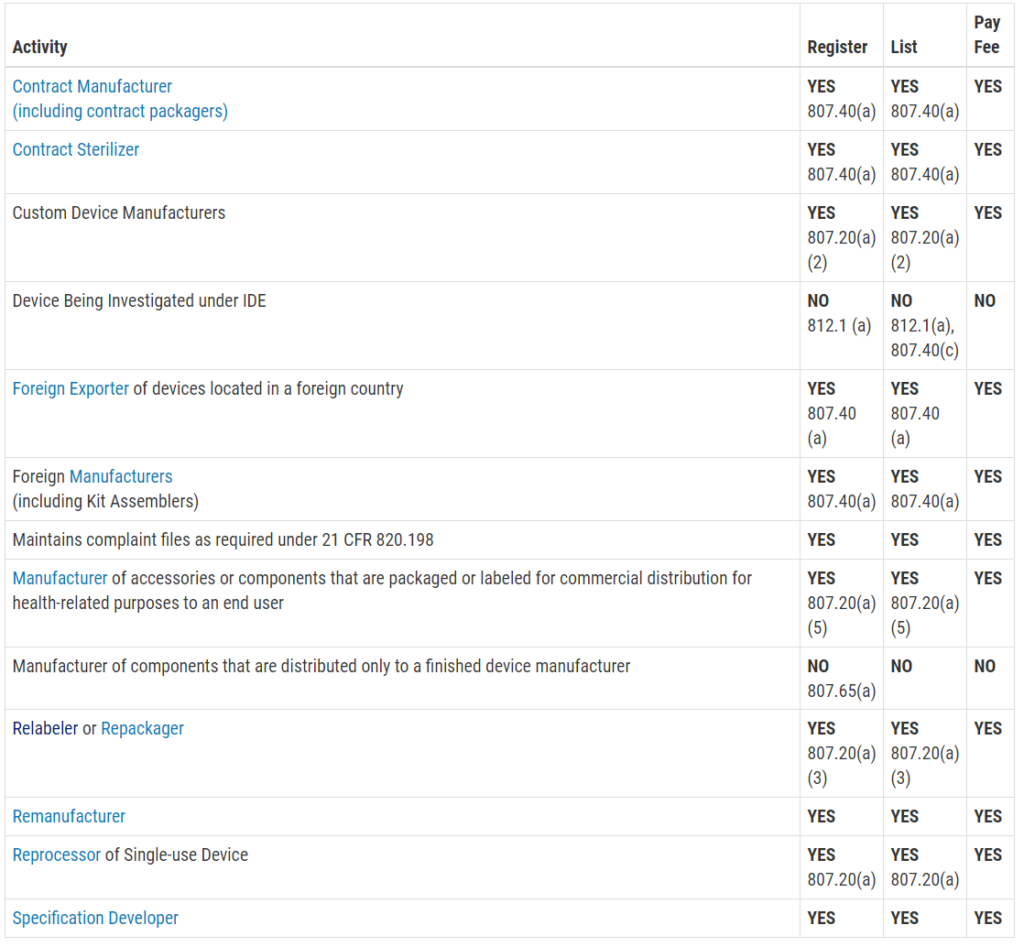
NSE Letter Cause #1: You failed to verify that the predicate is a legally marketed device.
If your predicate device is not legally marketed, you need to select a new predicate and resubmit. However, it is doubtful that your device would pass the refusal to accept the (RTA) screening process if the predicate was not legally marketed. If your predicate was not registered and listed with the FDA (check using this link), then you should have submitted a pre-sub request to determine if the agency has any problem with using the device you chose as a predicate. This is an essential question if the manufacturer is no longer in business, and the product is no longer for sale.
NSE Letter Cause #2: You failed to evaluate the substantial equivalence of your device’s intended use with the predicate.
The intended use of your predicate device is documented for every potential predicate since February 1992 on FDA Form 3881–which you can download along with the 510k clearance letter for the predicate. There is also an intended use documented for every device category in the applicable regulation for that device. This intended use is more generic than FDA Form 3881, but both are applicable. The FDA Form 3881 you submit for your device must be equivalent. I recommend a point-by-point comparison with regard to the following elements: 1) OTC vs. prescription use, 2) user, 3) patient population, 4) illness or medical condition, 5) duration of use, 6) environment of use and 7) target part of the body. Any difference can raise new issues of risk and may result in an NSE decision. However, the FDA typically will work with the company to modify the wording of FDA Form 3881 to ensure the intended use is equivalent or to make sure you provide clinical evidence to address the differences. In my pre-submission requests, I include a comparison document for the intended use to ensure that the FDA is aware of any differences in the intended use.
Cause #3: You failed to convince the FDA that technological differences do not raise different questions of safety and effectiveness.
Unless your device is identical in every way to the predicate device, you will have to persuade the FDA that differences do not raise questions of safety and effectiveness. At the beginning of the 510(k) process, it is helpful to document technological differences systematically. Specifically, this should include: 1) materials, 2) design, 3) energy source, and 4) other features. For each difference, you must justify why the difference does not raise different issues, or you must provide data to prove it. It is also possible that you were not aware of questions of safety and performance raised by technological differences. To avoid this problem, you can submit a detailed device description and draft labeling to the FDA in a pre-sub meeting request. If you ask questions about differences in a pre-sub meeting, you can avoid an NSE letter.
Cause #4: You failed to provide data demonstrating equivalence.
For each difference, you should determine an objective method for demonstrating that the difference is equivalent in safety and performance to the predicate. Your test method can be proposed to the FDA in a pre-sub request before testing. The FDA sees more than 3,000 companies propose testing methods to demonstrate equivalence each year. They have more experience than you do. Ask them in a pre-sub before you test anything. There may be a better test method, or you might need to adjust your test method. Sometimes results are unclear, but there might be another test you can perform to demonstrate equivalence, and then you can resubmit your 510k. Possibly you were unaware of the need to perform a test, and you were unable to complete a test within the 180 days the FDA allowed for submitting additional information. The good news is you now have all the time you need.
What is similar between all four causes of the NSE letter?
In all four root causes identified above, you could benefit greatly from the pre-sub meeting. Now you have an NSE letter, and you know which of the four reasons why your submission did not result in 510(k) clearance. However, the correction to your NSE letter may not be clear. Therefore, you should consider requesting a pre-sub meeting as quickly as you can. Most companies choose not to submit a pre-sub meeting request because they don’t want to wait 60-75 days. However, sometimes pre-sub meetings are scheduled sooner. In addition, 60-75 days is not as costly as receiving a second NSE letter.
Prevent a future NSE letter by requesting a pre-sub meeting
Regardless of your corrections for the current NSE letter, you should prevent future occurrences by planning to submit a pre-sub meeting request for every submission. I try to help clients gather all the information they need without a pre-sub meeting, but each new 510k reminds me why a pre-sub meeting is so valuable. You always learn something that helps you with the preparation of your 510k.
Help with Pre-sub meeting requests
The FDA published a guidance document for pre-sub meeting requests. If you need additional help, there is a webinar on this topic.
NSE letter: A CAPA plan for your 510k process Read More »