Medical Device Academy’s ISO 14971:2019 risk management training webinar is being expanded from a single webinar to a two-part webinar.
What’s new in this risk management training webinar?
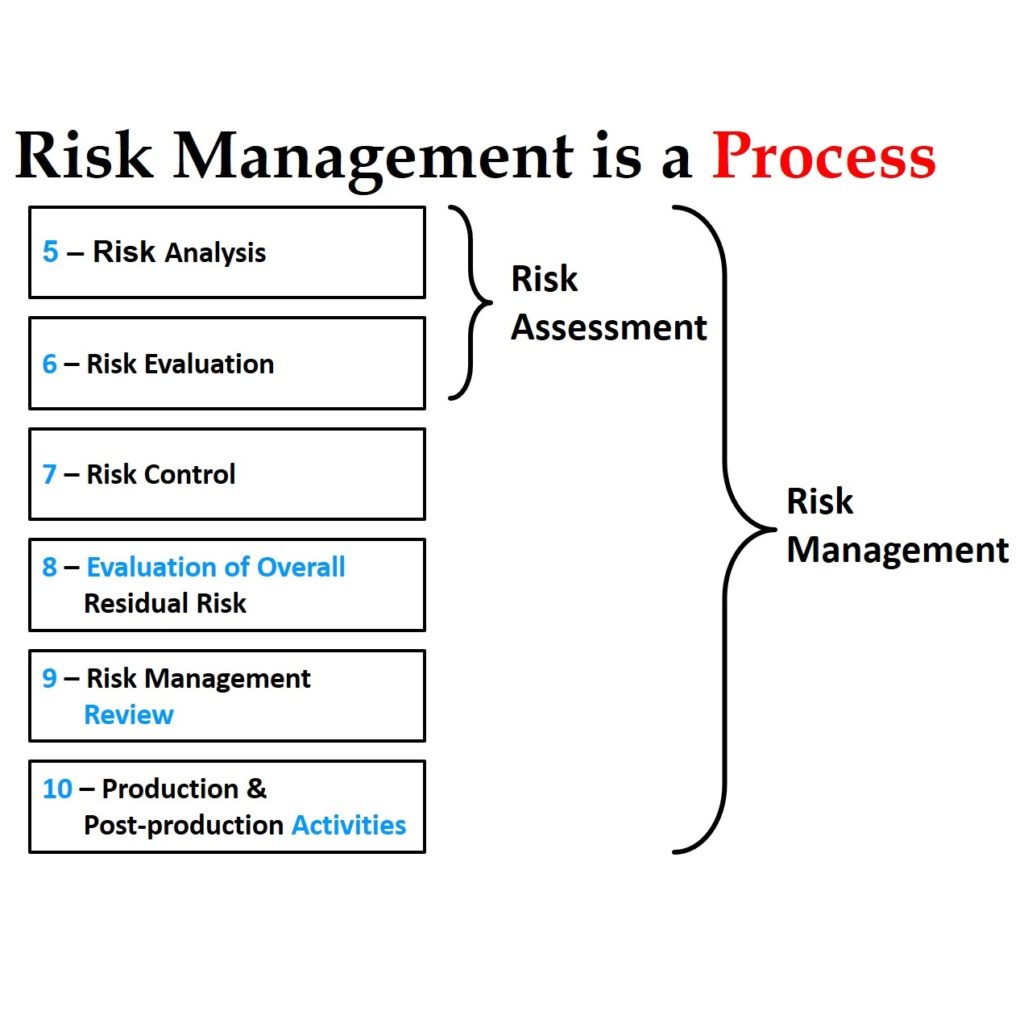
Our previous version of the ISO 14971 risk management training webinar was recorded on October 19, 2018. Although that webinar is 100% compliant with the 2019 version of the ISO 14971 standard, everyone considering the purchases of that webinar asks me to confirm this. The webinar was possible because we were using the draft version of the standard for the webinar. Over time we observe clients struggling with the implementation of the risk management process. It is a complex process, and covering the topic in a single webinar is not really feasible. Therefore, we now provide additional training webinars on hazard identification and benefit-risk analysis.
When is the risk management training webinar?
This webinar was recorded and you will receive access to the content as a Dropbox link. You will also receive the native slide deck. Any person that completes our training quiz will receive a training certificate, a corrected quiz, and the answer key to our quiz.
Register for the Risk Management Training Webinar for $129.00 (USD)
A risk management procedure compliant with ISO 14971 is the best practice for meeting the requirement in ISO 13485:2016, Clause 7.1. If you are unfamiliar with the ISO 13485 standard, please visit our page on “What is ISO 13485?“
VIEW OUR RISK MANAGEMENT PROCEDURE
CLICK HERE OR IMAGE BELOW:
About Your Instructor
Rob Packard is a regulatory consultant with ~25 years of experience in the medical device, pharmaceutical, and biotechnology industries. He is a graduate of UConn in Chemical Engineering. Rob was a senior manager at several medical device companies—including the President/CEO of a laparoscopic imaging company. His Quality Management System expertise covers all aspects of developing, training, implementing, and maintaining ISO 13485 and ISO 14971 certifications. From 2009 to 2012, he was a lead auditor and instructor for one of the largest Notified Bodies. Rob’s specialty is regulatory submissions for high-risk medical devices, such as implants and drug/device combination products for CE marking applications, Canadian medical device applications, and 510k submissions. The most favorite part of his job is training others. He can be reached via phone at +1.802.281.4381 or by email. You can also follow him on YouTube, LinkedIn, or Twitter.




