Which changes are forgotten in your MDR labeling procedure?
Did you forget any of the MDR labeling procedure requirements when you were updating your device labeling for CE Marking?
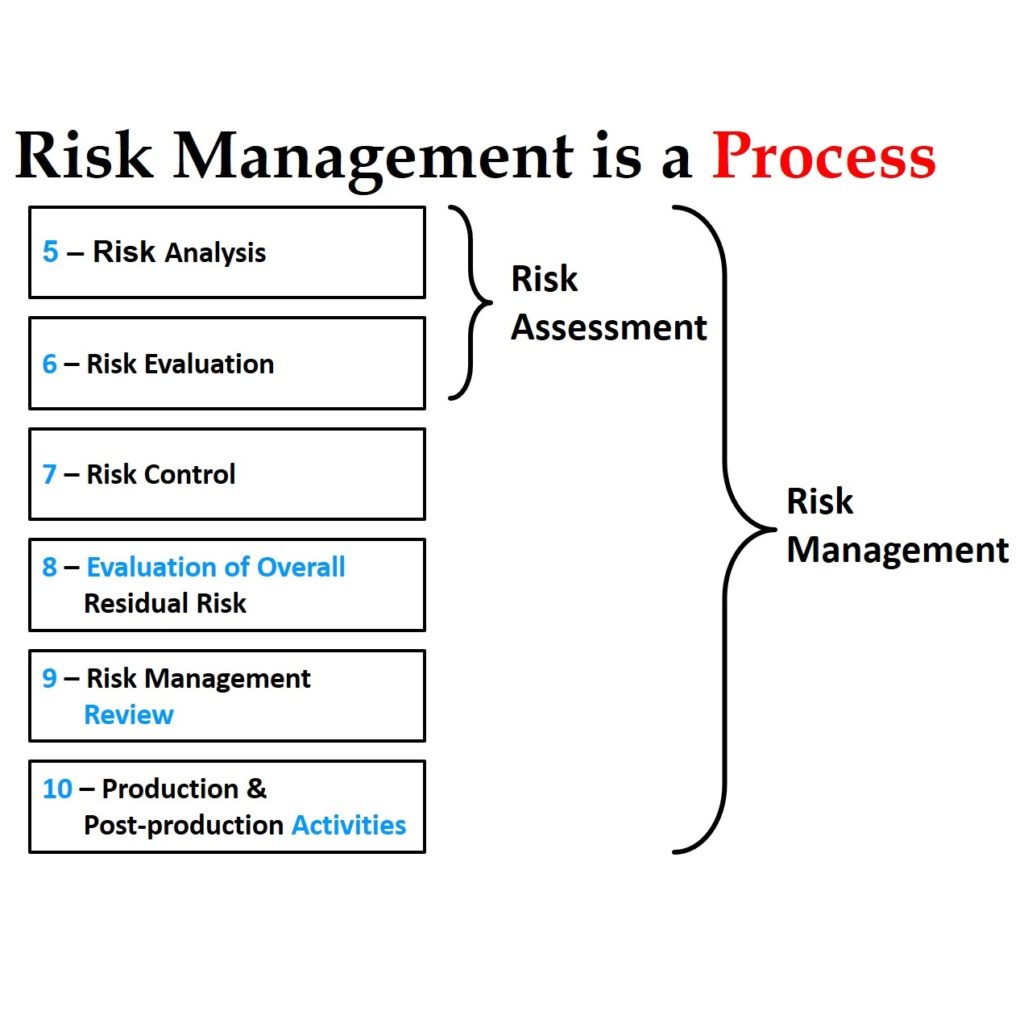
MDR Labeling Procedure
Don’t forget to subscribe to our YouTube channel for more medical device quality and regulatory training. The topic of this article is how to create an MDR labeling procedure for compliance with Regulation (EU) 2017/745 (MDR) for CE Marking of medical devices. The MDR does not actually include a requirement for a labeling procedure. In fact, the MDR doesn’t even specifically require that you have ISO 13485:2016 certification. ISO 13485:2016, clause 7.5.1 states that you shall implement “defined operations for labeling and packaging,” but the standard doesn’t specifically say that “the organization shall document procedures” for labeling. In 21 CFR 820.120, the FDA states that “each manufacturer shall establish and maintain procedures to control labeling activities.” But there is no similar requirement in the MDR.
MDR Quality System Requirements
Article 10 is the section of the MDR that defines the obligations for device manufacturers to create quality system procedures, but a labeling procedure is not specifically mentioned. Article 10(9)(a) states that your quality system shall include “a strategy for regulatory compliance, including…procedures for management of modifications to the devices covered by the system,” and this would include label changes and other control of other design changes. The next paragraph states that your quality system shall include, “identification of applicable general safety and performance requirements.” The general safety and performance requirements (GSPRs) are found in Annex I of the MDR, and the very last GSPR (i.e. GSPR 23) is for your label and instructions for use.
Then, which changes do you need to make for the MDR labeling procedure?
The GSPRs in Annex I of the MDR are longer than the Essential Requirements that were in the MDD. In addition to the new requirements for UDI compliance (which you should address in a UDI Requirements Procedure), GSPR 23 has new general requirements (i.e. 23.1) and new requirements for information on the sterile packaging (i.e. 23.3). There is also a more detailed specification for the information on the label (i.e. 23.2) and the information in the instructions for use (i.e. 23.4). The approach for demonstrating compliance with the GSPRs suggested in the MDR is to provide a checklist. Therefore, most manufacturers of CE Marked devices have replaced their Essential Requirements Checklist (ERC) with a GSPR checklist. However, if you are reviewing a draft label for approval, you don’t want to review and update your entire 22-page, GSPR checklist for every label.
The more efficient approach is to create one or more labeling checklists that are specific to the requirements in GSPR 23. If you create a separate checklist for the label, the information on the sterile packaging, and for the information in the instructions for use, then you would have three shorter checklists to complete. The label checklist and the checklist of the information on the sterile packaging would be only one page each, while the checklist for the instructions for use would be approximately four pages. There may be additional labeling requirements for specific countries and types of devices. Electrical medical equipment also has specific labeling requirements in IEC 60601-1 and IEC 60601-1-2. You will also need to create a user needs specification that can be used as criteria for summative usability testing (i.e. validation that the design and risk controls implemented meet the user needs specification). You should also document a use-related risk analysis (URRA), and perform formative testing, in order to identify critical tasks which need to be in the instructions for use to prevent use errors.
Are there any other MDR requirements that you should address in a labeling procedure?
There are two other requirements that should be addressed in your labeling procedure. The first is the general labeling requirements in GSPR 23.1. Withing GSPR 23.1, there are actually nine “sub-requirements.” The first “sub-requirement” in GSPR 23.1 is to provide the identity of the device, your company, and any safety and performance information needed by the user on the packaging or the instructions for use, and on your website. Many manufacturers do not want to make this information available on their website, because it makes it easier for competitors to copy the instructions for use, but this is not optional. This requirement and the other eight requirements in GSPR 23.1 could be included in your procedure or as part of a fourth labeling checklist associated with your MDR labeling procedure.
The second requirement is the requirement to translate your instructions for use into an official Union language(s) determined by the member state where your device will be made available to the intended user or patient. Creating these translations, and verifying the accuracy of the translations, can be expensive and burdensome–especially if your device is sold in most of the member states.
You might also consider implant cards as labeling requirements and try to add them to your MDR labeling procedure. However, if the requirement for implant cards (see Article 18 of the MDR) is applicable to your company you should create an implant card procedure instead because this is a detailed and critical requirement that will not apply to most of the other labels in your company. You should make sure that the implant card procedure is compliant with MDCG 2021-11 released in May 2021 and MDCG 20201-8 v2 release in March 2020. These guidance documents also have great examples of how to design your implant cards.
Other changes in labeling requirements
The ISO 15223-1:2016 standard has been revised and was expected for release at the end of 2020. However, only draft versions are currently available (i.e. ISO/DIS 15223-1:2020). This new version of the standard for symbols to be used with labeling will also need to be updated shortly in your MDR labeling procedure. This new version is already referenced in the medical device standard for information provided by the manufacturer (i.e. EN ISO 20417:2021)–which supersedes EN 1041:2008. Consultants and chat rooms have argued over whether the requirement for identifying the importer must be on the label or if it could be presented in other documents. EN ISO 20417:2021 resolves this dispute in section 7.1: “Where necessary, the label of a medical device or accessory shall include the name or trade name and full address of the importer to which the responsible organization can refer.” In the note following that clause, it clarifies that “This can be required by the authority having jurisdiction.” There is even a new symbol referenced for importers (i.e. Symbol 5.1.8 in ISO 15223-1).
If you have specific questions about device labeling or MDR compliance, please use our calendly app to schedule a call with a member of our team. You can also purchase our labeling and translation procedure (SYS-030) to save yourself the time and effort of making your own versions of the labeling checklist described above.
About the Author

Robert Packard is a regulatory consultant with 25+ years of experience in the medical device, pharmaceutical, and biotechnology industries. He is a graduate of UConn in Chemical Engineering. Robert was a senior manager at several medical device companies—including the President/CEO of a laparoscopic imaging company. His Quality Management System expertise covers all aspects of developing, training, implementing, and maintaining ISO 13485 and ISO 14971 certification. From 2009-2012, he was a lead auditor and instructor for one of the largest Notified Bodies. Robert’s specialty is regulatory submissions for high-risk medical devices, such as implants and drug/device combination products for CE marking applications, Canadian medical device applications, and 510(k) submissions. The most favorite part of his job is training others. He can be reached via phone 802.258.1881 or email. You can also follow him on Google+, LinkedIn or Twitter.
Which changes are forgotten in your MDR labeling procedure? Read More »