Instructor engagement is the secret to training effectiveness
The author discusses his personal experience with quality and regulatory training and shares his secrets for better instructor engagement.
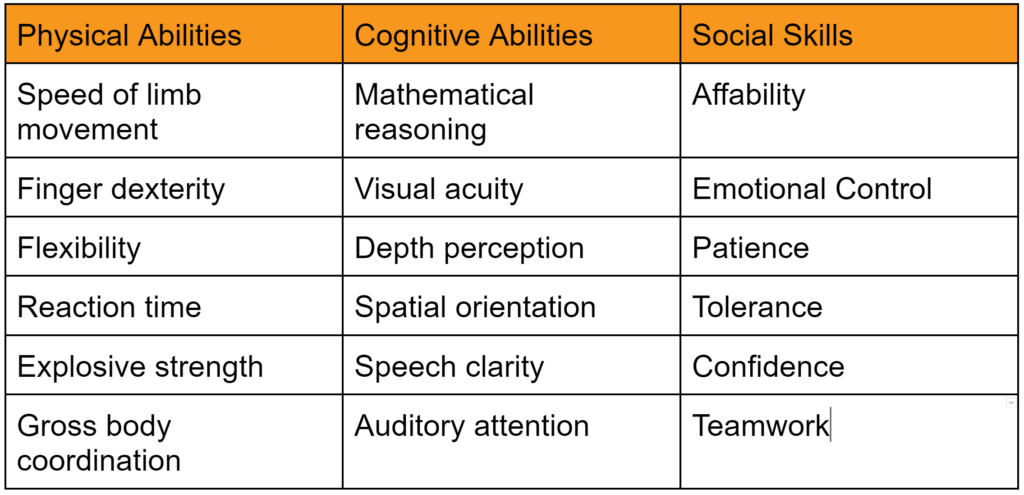
What is instructor engagement?
Instructor engagement is a term that describes actions taken by the instructor to involve their class, whether the training is on-line or in person. Instructor engagement includes six basic elements:
- Training content and format
- Verbal communication
- Non-verbal communication
- Training environment
- Audience
- Audience involvement
Any one of the above six elements can ruin a training class, but a great instructor can compensate for weaknesses in any one area by taking advantage of the other elements.
To be successful, you need to hook your audience in the first ten seconds
It is crucial to engage your audience in the first ten seconds. When the audience is live, if you don’t engage them immediately they will find something on their phone to distract them. If the audience is on-line, they will swipe to the next video in their feed. Toastmasters suggests beginning your presentation by using one of five methods for hooking your audience:
- Tell a story
- Make a bold statement
- Ask a question
- Get the audience to laugh
- Ask the audience to visualize something

Consequences of poor engagement
If an instructor does not engage students, the best case scenario is that the training will not be effective. In other words, the class will not learn the material being taught or they will retain the information for less than 24 hours. For the student, their time and money was wasted. For the instructor, they will feel exhausted at the end of the training and they will have trouble finding future training jobs.
Results of good instructor engagement
If a class is engaged in training they will learn the material, retain the information in their long-term memory, and the will recommend the instructor to other people that are interested in learning about the topic. For the student, their time and money was well spent. For the instructor, they will feel energized at the end of the training and students will come up to them at the end of the training asking for a business card and to discuss future training opportunities.
Where can you find an examples of good and bad instructor engagement?
When I first wrote a blog on this topic it was 2012 and there were very few blogs and almost no videos dedicated to quality systems or regulatory affairs. Twelve years later, almost nobody reads blogs and there are multiple competitors that publish new videos weekly. The primary channel for watching videos is YouTube, but YouTubers are simultaneously live-streaming on YouTube, LinkedIn, and Facebook. These channels are the best place to find examples of good and bad instructor engagement. You will probably have a strong opinion about the quality of the speaker in the first ten seconds, but the algorithms that guide your surfing of these platforms will automatically steer your viewing to the best videos. These videos are not considered to be the best solely on content and format. The amount of audience engagement is the biggest driver. The algorithm recommends videos based upon the percentage of audience retention, the number of comments by viewers, and the number of people that share the link to your video.
Why doesn’t Dr. Shulman’s video rank higher in the algorithm?
The video I embedded in this post has only 554 views currently, but it has been posted on YouTube almost six years. Why doesn’t this video rank higher and get recommended by YouTube to more people? Because there is no involvement of the original audience or the current YouTube audience. Despite the talented speaker and the use of video with clear PPT slides, there are no comments on YouTube. The content and format is good, and the verbal communication is good. However, RAPS has Dr. Shulman standing behind a podium at the event so the non-verbal communication is not as strong as it could be. In addition, large conferences are typically one of the weakest environments for encouraging instructor engagement with the audience. The video could receive much higher rankings if the original audience was involved in the video as well. If the question and answer session were included, that might have helped. RAPS could also improve the video’s performance by adding more details to the description of the video. Finally, the performance of the video is impacted by the number of subscribers to the RAPS YouTube channel and the engagement of those subscribers. To demonstrate this, I will add a comment to the video. That should cause a small increase in engagement and viewers.
How can you improve your internal quality system training?
Anyone can read and understand a procedure, but this is the least effective method of training people. You could also have employees watch a training video, but quality assurance and regulatory affairs are among the most boring topics on planet earth. Most of the training out there is “Blah, blah, blah…” and “Death by PowerPoint.” Instructor engagement for that type of training is poor, and it could get you fired. Don’t read your slides, don’t turn your back on the audience (or they’ll attack), and PLEASE don’t ever ask someone to read the definition of nonconformity out loud to the rest of the group. Inspire and engage the class. You need to get your audience to pay attention, ask questions, and share their own thoughts out loud. For example, instead of using a PowerPoint, try displaying the actual procedure and ask an audience member to find each of the points you are teaching in the procedure. You could even teach them a cool search tool (i.e., CTRL + F) to find the content. You might even adding a symbol to the procedure to help them find those requirements.
Nine ways to improve your own instructor engagement
If you are hiring a consultant to help you with quality and regulatory training, then you certainly want to hire an expert. However, it is more important that the speaker is engaging. Knowing this fact, you could try improving your own presentation skills to achieve higher instructor engagement for a lot less money. I’m six-foot, six inches tall, and I have a loud booming voice. My mother has red hair, and she was an opera singer. I’ve got the voice to fill any auditorium and stage presence to match. But you don’t have to be big, tall, or loud to capture the attention of your audience. Here are my top nine ways to improve instructor engagement:
- Practice vocal variety
- Move, don’t stay stationary
- Ask the audience questions
- Use anecdotes, case studies, and stories
- Try using props
- Take breaks
- Plan a surprise
- Force feed the audience legal stimulants
- Give students homework
What is vocal variety, how does it impact instructor engagement?
Vocal variety is more than just the volume of the speaker. Vocal variety consists of pitch, tone, volume, and pace. Generally I speak too fast and my voice is very loud. Therefore, if I want to emphasize a point I can exercise two changes in my voice to immediately capture the attention of students: 1) speak softly, and 2) speak slowly. Another approach I have used, is to speak slowly and repeat myself, but the most dynamic way to get your audience’s attention is to stop speaking for a few seconds. Silence is powerful.
Many people struggle to understand how to vary their pitch, but they’re overthinking it. When we ask a question, we raise the pitch of our voice slightly at the end. This upward inflexion of our voice signals to listeners that we are asking a question. For example, if you repeat the last one to three words in the other person’s sentence, and you say this with an upward inflection, the listener will perceive that you are asking a question to better understand what they mean. This is much more effective than asking “what do you mean?” For example, if a student in the class says “Is it enough if we perform annual reviews?” As the instructor you can employ the technique of mirroring by asking, “Annual reviews?” This will encourage the other person to elaborate on what they meant by “annual reviews.” This technique ensures that you know what the student meant, and it gives you time to consider what they said before you respond. This is also a strategy recommended by expert negotiators.
Movement is attractive
Most of our brain power is dedicated to processing what we see–not what we hear. Therefore, listeners are more likely to notice when you move. You can jolt your audience awake simply by stepping out from behind a podium or changing your movement pattern (e.g., occasionally moving forward instead of pacing side to side). Movement also includes body language. You can modify your posture, stance, and position in front of the audience to communicate information non-verbally. You can use gestures to communicate non-verbally or you can use facial expressions. For example, if you frown and shrug your shoulders, what does that mean?
Questions are essential for instructor engagement
No matter how smooth and eloquent your voice is, nobody wants to hear only you speak. This is one of the reasons conferences have multiple speakers. However, during a single presentation you can get students to participate and present some of the information by asking them questions. There are a few techniques that help get the audience to speak more. Instead of asking a question to the group as a whole, try pointing to a specific person, ask them their name, and then ask them a question. Second, in order to “break the ice” at the end of your presentation, have some “seed questions” prepared. If you are conducting a webinar, seed questions can be read by you. In a live presentation, you can give a list of seed questions to your host or friends in the audience. Finally, you can also begin your training with a question (i.e., Toastmaster hook #3 above).
Tell a story and make a point
For each teaching point you should consider using an anecdote, case study, or a related story. The audience will want to know how the story ends, and they are more likely to remember the story. If you haven’t conducted more than 1,000 audits, traveled all over the world, or have more than 25 years of experience–don’t despair. You can always interview other people on any topic to get their stories. If you are looking for tips on how to construct a story, Toastmasters comes to my rescue again. Here’s their recommended six-part structure for the Hero’s Journey:
- Setup
- Inciting incident
- Progressive complications
- Insight
- Climax and resolution
- Lesson
What kind of prop can you use for quality and regulatory?
In one of the paragraphs below, I mention a simple prop that you can use for training–product samples. I did this in one of our live-streaming YouTube videos where I explained how to review medical device labeling. But you can use other things in your environment. For example, the first public speaking course I ever had was taught by a man with Polio that used crutches. He used one of his crutches as a prop to demonstrate how he looked over a fence.
How often should you take breaks?
Students cannot maintain a state of alertness and attention indefinitely. Your body naturally cycles between higher and lower alertness every 90 minutes. After 90 minutes it becomes harder to focus and you need to take a break. If you can, splitting 90 minutes into two 45-minute sessions is even better. You can also experiment with two strategies for better instructor engagement: 1) conduct a “pop quiz” after a break to make sure the audience understood the information they were just taught, and 2) don’t be afraid to adjust the breaks slightly to coincide with a change in topic. Changing topics at a break allows you to repeat the most important points three times. The first time when you introduced a topic, the second time when you have concluding remarks at the end of a topic, and a third time after the break when you make sure the audience understood the material you presented before the break.
What kind of surprise will engage your audience?
There are two strategies for using a surprise: 1) promise to surprise them in the future, or 2) don’t tell anyone until you surprise them. The first strategy works best when you are trying to get people to watch until the end of the training. This is commonly used by YouTubers to get people to watch the video until the end. Unfortunately, this backfires because we can fast-forward to the end. I’m not suggesting that you shouldn’t surprise your audience, but the surprise needs to delight your audience. You might also need to surprise and delight your audience more than once. Even then, some of your audience will still nod off and completely ignore you. When this happens, throw a Snickers bar at the offending student.

How to force-feed students legal stimulants
If legal counsel recommends against using projectiles to encourage class participation, you might also consider one of my all-time genius ideas–consuming dangerously large quantities of caffeine. I was scheduled for a two-day course in Ottawa, but the day before I needed to perform an audit in Pennsylvania. My flight was the last flight into Ottawa, which arrived at approximately 1 o’clock in the morning. My arrival was delayed an additional hour in customs by the person in front of me who was trying to smuggle an extra carton of smokes into the country. Just before 4 a.m., my taxi arrived at the Albert at Bay Suite Hotel. The class started at eight in the morning. I made it to class on time, and the excessive consumption of several pots of black coffee helped get me to lunch. Then my legs started getting a little shaky. Fortunately, there was a convenience store next door that sold my favorite chocolate–the Dark Aero bar! After four of these monstrous doses of cacao, and another pot of coffee, I could have listened to the lecture on the Canadian Medical Devices Regulations all night. The only problem is that my hands are still shaking 15 years later.

Why was the instructor engagement high in Ottawa?
Despite the physical handicap of sleep deprivation, I still learned a ton from my course in Canada. Here’s why:
- The instructors were both regulatory experts that were able to share anecdotes, case studies, and stories about real-world application of the Canadian Medical Devices Regulations. One of the instructors even worked for Health Canada.
- The audience was hyper-motivated to pass the course, because everyone in the class worked for a Notified Body that had sponsored them to take the course. In order to stay employed and get a raise, I needed to pass that course. If I failed the exam, I had to absorb the cost to travel back to Ottawa and retake the course in February (BRRRR!).
- The instructors brought more than a dozen medical devices to the class. These props gave us something to read, touch, and ask questions about. The instructors broke us up into small teams to study the labeling and instructions for use of each device. Even students from Japan, Europe, and Australia were familiar with some of the products. This was critical because we all needed to be able to identify incorrect Canadian labeling.
- The best instructor engagement tool used was humor. The instructor from Health Canada was hilarious. He had everyone laughing at his jokes for the entire course. Most of the jokes were not funny enough for a stand-up routine, but this was a mandatory regulatory course on Canadian regulations. Who would even expect a chuckle? Despite the strengths of these instructors, there is only one reason why I know the Canadian Medical Devices Regulations (CMDR), as well as I do. I use them every single week.
When students are forced to do homework, they will pay attention
After completing my CMDR training, I had to audit 162 days for BSI in 2011. Ninety percent of those 162 days were for companies that required a Canadian Medical Device License. This forced me to use the information I learned in the course. I was also consulting for companies at the same time I was auditing for BSI. Consulting clients hired me to prepare and submit the Canadian Medical Device License Applications for them. I also had to create procedures for Canadian Licensing, Incident Reporting, and Recalls. I spent another 60+ days in 2011 doing consulting, which helped me hone my knowledge of Canadian device regulations.
Teaching others will make you a guru
Most people are terrified to speak on any topic–even to a small group of coworkers. However, I believe that teaching others is the secret to becoming a guru on any topic. I was one of BSI’s instructors that taught the regulatory comparison course from 2010 to 2012, which compared the regulations of the USA, Canada, Europe, Australia, and Japan. Therefore, at least once a month, I had a classroom of 6-20 people asking me challenging questions about how to interpret and apply regulations from each of these countries to their products. I used every bit of knowledge I learned in that course in Ottawa, and I started using that knowledge immediately after the course. Peers, clients, and students challenge my knowledge of these topics every day. This is what makes you a subject matter expert. If you need to learn something about quality assurance or regulatory affairs watching a one-hour webinar, reading a blog, taking a five-day course, or shadowing another more experienced person is not enough. In the end, all of the above will get you to the level of barely competent! If you want to master any topic, you need to practice instructor engagement and use everything you learn for several years.
Instructor engagement is the secret to training effectiveness Read More »