FDA User Fees for FY 2026 released on July 31, 2025
The FDA User Fees for FY 2026, October 1, 2025 – September 30, 2026, were released on Thursday, July 31, 2025.
What are FDA User Fees?
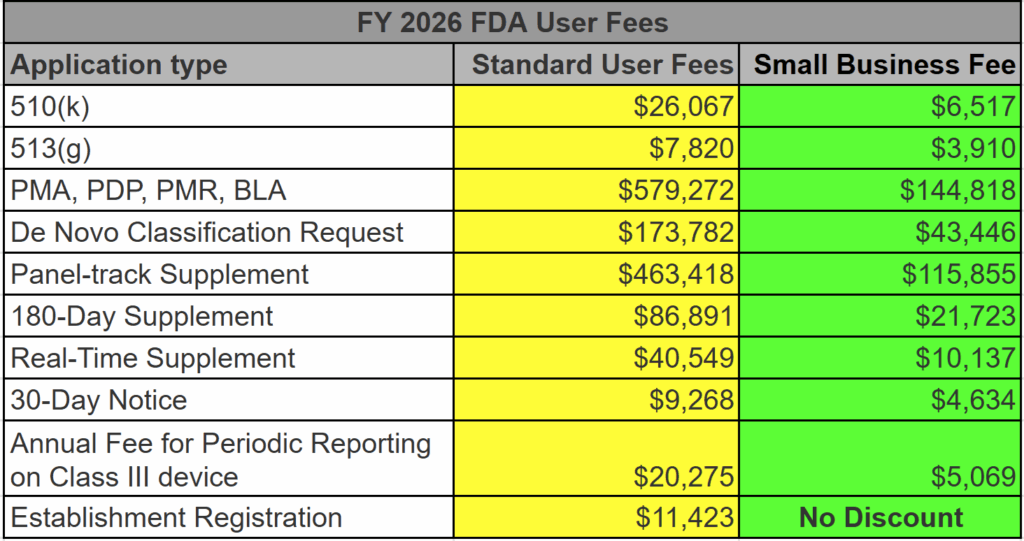
At the very core of it, the FDA user fees fund the FDA Office of Device Evaluation (ODE) budget. Without these user fees, the FDA cannot begin reviewing a medical device submission. This includes 510k, PMA, and De Novo submissions. Before the FDA assigns a reviewer to your submission, you must pay the appropriate device user fee in full unless eligible for a waiver or exemption. If you pay the user fee by credit card, you must allow a few extra days for the user fee to clear. Otherwise, your submission will be placed on “User Fee Hold.” Small businesses may qualify for a reduced fee. The FDA announced the FY 2026 FDA User Fees on July 31, 2025. The FDA will announce the user fees for FY 2027 in a Federal Register notice next August 2026.
What are the FDA User Fees for FY 2026?
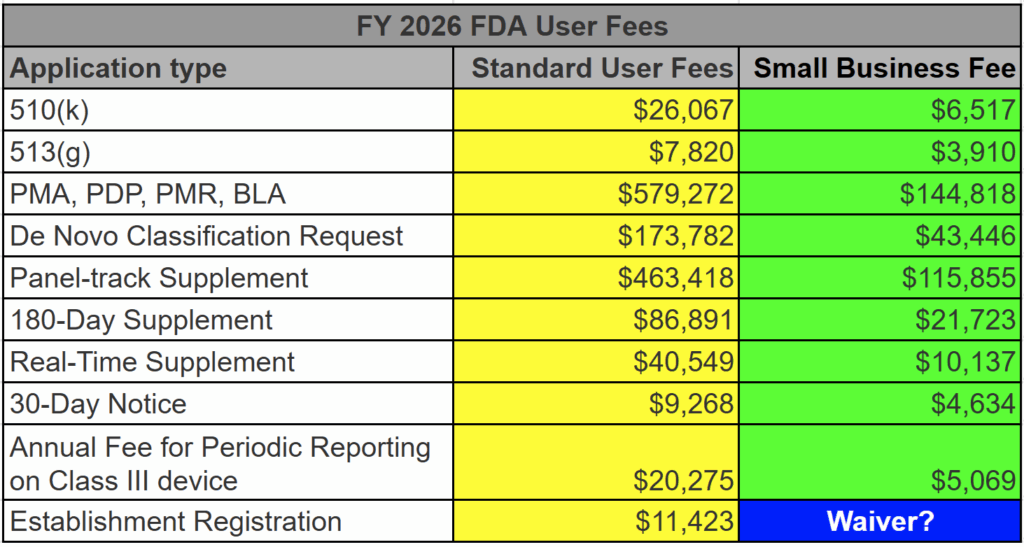
Is there a small business discount for the establishment registration?
Certain small businesses certified through the small business designation (SBD) program may qualify for a waiver for the registration fee if the business and its affiliates:
- have gross receipts and sales of no more than $1 million USD,
- can demonstrate paying the registration fee would represent a financial hardship (for example, proof the business is in active bankruptcy), and
- has proof of a prior year’s payment of the registration fee.
How much did user fees increase for FY 2026?
The increase in FDA user fees from FY 2025 to FY 2026 was 7.12%, except the annual FDA Registration fee, which increased by 23.1% to $11,423. There are three components to the increase:
- Base Fee = a statutory base fee for each FDA user fee
- Standard Fee = an inflation-adjusted statutory base fee
- Adjusted Fee = adjusted fee to meet revenue target
The reason for each component for the user fees is described in the Federal Register.
When does the FY 2026 increase take effect?
Each year the new FDA user fees take effect on the 1st day of the FDA’s new fiscal year (i.e., October 1). You cannot pay the annual registration fee for FY 2026 until October 1, 2026, and the last day you can submit under the FY 2025 user fee pricing is Tuesday, September 30, 2025. For the submission to be accepted under the current fiscal year, the submission must be uploaded to the Customer Collaboration Portal (CCP) no later than 4:00 p.m. ET on the 30th.
What do you do if you have already paid the FY 2025 price?
If you already paid the FY 2025, and your submission is received after 4:00 p.m. ET on September 30, 2025, you must complete FDA Form 3914 for an FDA user fee payment transfer request. You will also need to pay the difference in user fees (i.e., 7.12%). If your submission is received before the FY 2025 user fee is transferred and you have paid the difference in user fees, your submission will be placed on a user fee hold. If you paid the FY 2025 user fee and are not ready to transfer your previously paid user fee to FY 2025 (and pay the difference), you can request an FDA user fee refund by filling in an online form.
What is the annual registration fee for FY 2026 due?
The annual establishment registration user fee can be paid any time between October 1 and December 31. If you pay late, there is no penalty, but your registration status will be inactive, and you cannot submit new device submissions or import products to the USA. If you are not yet distributing any devices in the USA, you are not required to have your establishment registered, and establishment registration is not required before submitting a new device submission. If you are not required to register yet, when you are paying the user fee for a new device submission on the Device Facility User Fee (DFUF) website, you will click the “Yes” button because there is no “N/A” option for the question below.
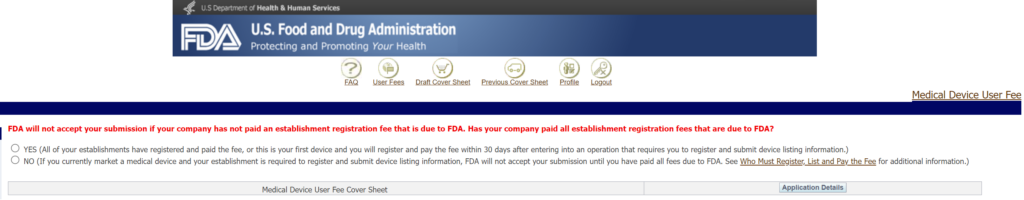
Is the annual FDA registration fee prorated?
Annual registration payments are not prorated when you are paying in the middle or even near the end of the year for your initial registration. Therefore, you will need to consider if the revenues you expect to gain before the end of the current fiscal year are worth the registration cost. If you need any help with annual registration or you need a US Agent, we offer these consulting services.
FDA User Fees for FY 2026 released on July 31, 2025 Read More »